US fluoride supplements: The US Food and Drug Administration narrowed the use of pediatric fluoride supplements, saying they are no longer recommended for children under 3 or for older children who are not at high risk of tooth decay. The move, part of Health Secretary Robert F. Kennedy Jr.’s broader scrutiny of fluoride, scales back guidance that previously allowed prescriptions for infants as young as six months.
Regulators stopped short of pulling the products from shelves, but issued warning letters to four manufacturers instructing them not to market outside the new limits. Fluoride tablets, lozenges and infant drops are typically used in communities with low fluoride levels in drinking water to reduce cavities.
Also Read | Removing fluoride from water could cause 25 million more cavities in US kids: Study
In a new scientific review released Friday, the FDA said supplements offer limited dental benefit and may be linked to emerging safety concerns, including gastrointestinal issues, weight gain and effects on cognition. The agency also cautioned that because fluoride can kill bacteria on teeth, it may also alter the gut microbiome, with potential broader health effects. A form letter was sent to dentists and other providers outlining the risks.
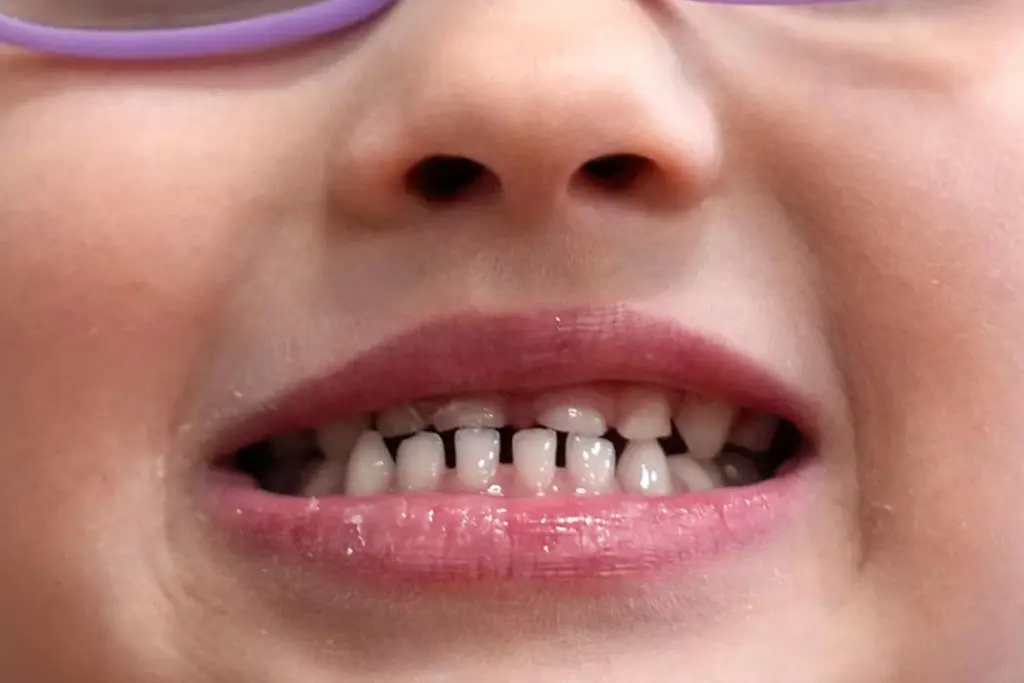
The American Dental Association disputed the concerns, maintaining that fluoride used as prescribed is safe, though the FDA noted the known downside of dental fluorosis, spotting or discolouration from excess fluoride. Dentists warned the restrictions could worsen cavity rates in rural areas without fluoridated water. Kennedy has also advocated ending water fluoridation nationwide.
According to the CDC, fluoride helps prevent cavities by remineralising teeth. The FDA said its action does not affect fluoride toothpastes, mouthwashes, or in-office treatments for adults or children.